Early
Early
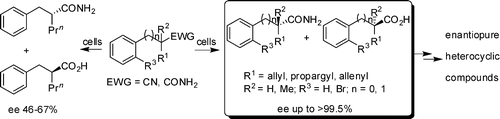
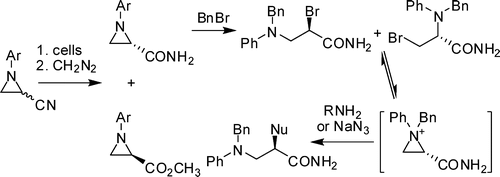
Nitrile and amide biotransformations for the synthesis of enantiomerically pure 3-arylaziridine-2-carboxamide derivatives and their stereospecific ring-opening reactions
Jin-Yuan Wang, De-Xian Wang, Jie Pan, Zhi-Tang Huang, Mei-Xiang Wang* J. Org. Chem. 2007, 72, 9391-9394.
.
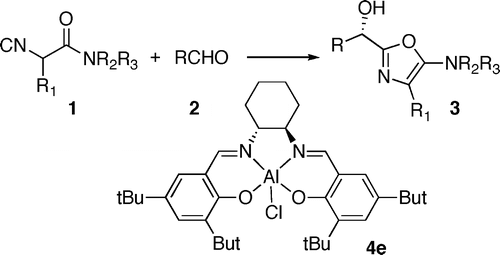
Asymmetric Lewis acid catalyzed addition of isocyanides to aldehydes – synthesis of 5-amino-2-(1-hydroxyalkyl)oxazoles
Shixin Wang, Mei-Xiang Wang,* De-Xian Wang, Jieping Zhu* Eur. J. Org. Chem. 2007, 4076-4080.
Chiral salen-aluminum complex as a catalyst for enantioselective α-addition of isocyanides to aldehydes: Asymmetric synthesis of 2-(1-hydroxyalkyl)-5-aminooxazoles
Shi-Xin Wang, Mei-Xiang Wang,* De-Xian Wang, Jieping Zhu* Org. Lett. 2007, 9, 3615-3618.
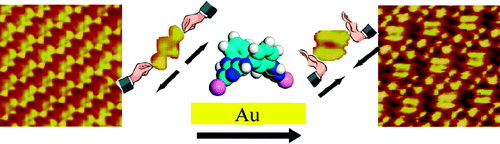
Adlayer structures of aza- and/or oxo-bridged calix[2]arene[2]triazines on Au(111) investigated by scanning tunneling microscopy (STM)
Cun-Ji Yan, Hui-Juan Yan, Li-Ping Xu, Wei-Guo Song, Li-Jun Wan,* Qi-Qiang Wang, Mei-Xiang Wang* Langmuir 2007, 23, 8021-8027.
Synthesis and structure of nitrogen bridged calix[5]- and -[10]-pyridines and their complexation with fullerenes
Shi-Qiang Liu, De-Xian Wang, Qi-Yu Zheng, Mei-Xiang Wang* Chem. Commun. 2007, 3856-3858.
Highly selective recognition of diols by a self-regulating fine-tunable methylazacalix[4]pyridine cavity: Guest-dependent formation of molecular-sandwich and molecular-capsule complexes in solution and the solid state
Han-Yuan Gong, De-Xian Wang, Jun-Feng Xiang, Qi-Yu Zheng, Mei-Xiang Wang* Chem. Eur. J. 2007, 13, 7791-7802.
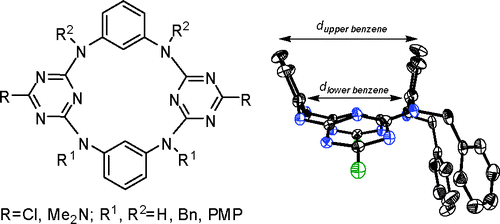
En route to inherently chiral tetraoxacalix[2]arene[2]triazines
Bao-Yong Hou, Qi-Yu Zheng, De-Xian Wang, Mei-Xiang Wang* Tetrahedron 2007, 63, 10801-10808.
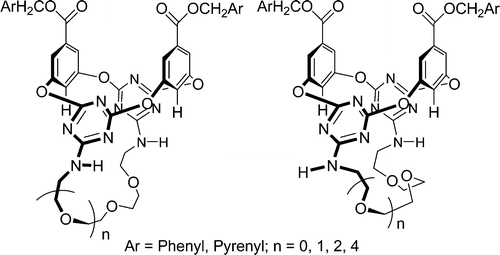
Synthesis and structure of upper-rim 1,3-alternate tetraoxacalix[2]arene[2]triazine azacrowns and change of cavity in response to fluoride anion
Bao-Yong Hou, De-Xian Wang, Hai-Bo Yang, Qi-Yu Zheng, Mei-Xiang Wang* J. Org. Chem. 2007, 72, 5218-5226.
Remarkable electronic and steric effects in the nitrile biotransformations for the preparation of enantiopure functionalized carboxylic acids and amides: Implication for an unsaturated carbon-carbon bond binding domain of the amidase
Ming Gao, De-Xian Wang, Qi-Yu Zheng, Zhi-Tang Huang, Mei-Xiang Wang* J. Org. Chem. 2007, 72, 6060-6066.
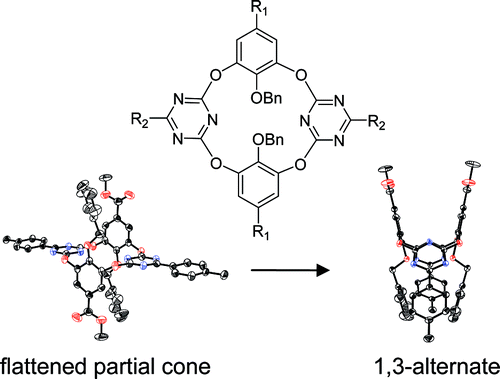
Formation and conformational conversion of flattened partial cone oxygen bridged calix[2]arene[2]triazines
Qi-Qiang Wang, De-Xian Wang, Qi-Yu Zheng, Mei-Xiang Wang* Org. Lett. 2007, 9, 2847-2850.
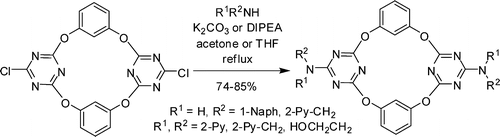
Efficient functionalizations of heteroatom-bridged calix[2]arene[2]triazines on the larger rim
Hai-Bo Yang, De-Xian Wang,* Qi-Qiang Wang, Mei-Xiang Wang* J. Org. Chem. 2007, 72, 3757-3763.
Separation and recognition of the enantiomers of trans arycyclopropanecarboxylic acids and their amide and nitrile derivatives on polysaccharide stationary phases
De-Xian Wang,* Xiao-Xia Zhang, Jin-Lei Zhang, Mei-Xiang Wang* Chromatographia 2007, 65, 343-348.
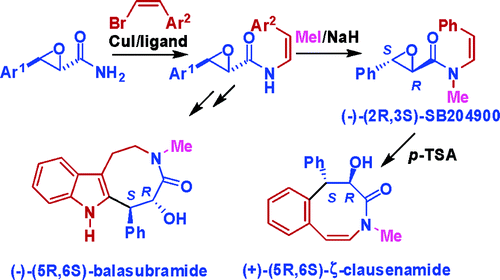
Highly efficient and stereoselective N-vinylation of oxiranecarboxamides and unprecedented 8-endo-epoxy-arene cyclization: Expedient and biomimetic synthesis of some clausena alkaloids
Luo Yang, Gang Deng, De-Xian Wang, Zhi-Tang Huang, Jie-Ping Zhu, Mei-Xiang Wang* Org. Lett. 2007, 9, 1387-1390.
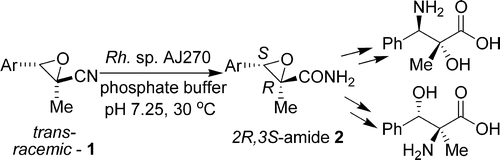
Nitrile biotransformations for the efficient synthesis of highly enantiopure 1-arylaziridine-2-carboxylic acid derivatives and their stereoselective ring-opening reactions
Jin-Yuan Wang, De-Xian Wang, Qi-Yu Zheng, Zhi-Tang Huang, Mei-Xiang Wang* J. Org. Chem. 2007, 72, 2040-2045.
Synthesis of tetraazacalix[2]arene[2]triazines: Tuning the cavity by the substituents on the bridging nitrogen atoms
Qi-Qiang Wang, De-Xian Wang, Hong-Wei Ma, Mei-Xiang Wang* Org. Lett. 2006, 8, 5967-5970.
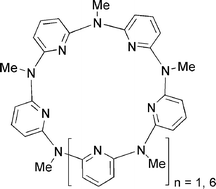
Methyazacalixpyridines: Remarkable bridging nitrogen-tuned conformations and cavities with unique recognition properties
Han-Yuan Gong, Xiao-Hang Zhang, De-Xian Wang, Hong-Wei Ma, Qi-Yu Zheng, Mei-Xiang Wang* Chem. Eur. J. 2006, 12, 9262-9275.
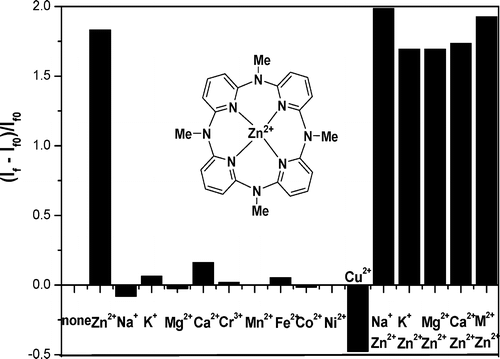
Methylazacalix[4]pyridine: En route to Zn2+-specific fluorescence sensors
Han-Yuna Gong, Qi-Yu Zheng, Xiao-Hang Zhang, De-Xian Wang, Mei-Xiang Wang* Org. Lett. 2006, 8, 4895-4898.
An unusual β-vinyl effect leading to high efficiency and enantioselectivity of the amidase, nitrile biotransformations for the preparation of enantiopure 3-arylpent-4-enoic acids and amides and their applications in synthesis
Ming Gao, De-Xian Wang, Qi-Yu Zheng, Mei-Xiang Wang* J. Org. Chem. 2006, 71, 9532-9535.
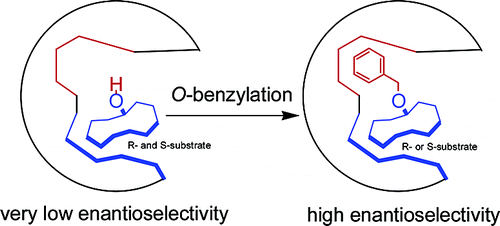
Dramatic enhancement of enantioselectivity of biotransformations of β-hydroxy nitriles using a simple O-benzyl protection/docking group
Da-You Ma, Qi-Yu Zheng, De-Xian Wang, Mei-Xiang Wang* Org. Lett. 2006, 8, 3231-3234.
Nitrile biotransformations for the practical synthesis of highly enantiopure azido carboxylic acids and amides, 'click' to functionalized chiral triazoles and chiral β-amino acids
Da-You Ma, De-Xian Wang, Qi-Yu Zheng, Mei-Xiang Wang* Tetrahedron: Asymmetry 2006, 17, 2366-2376.
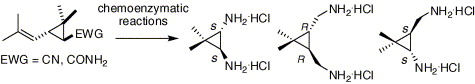
Chemoenzymatic synthesis of enantiopure geminally dimethylated cyclopropane-based C2- and pseudo-C2-symmetric diamines
Guo-Qiang Feng, De-Xian Wang, Qi-Yu Zheng, Mei-Xiang Wang* Tetrahedron: Asymmetry 2006, 17, 2775-2780.
Biocatalytic synthesis of highly enantiopure 1,4-benzodioxane-2-carboxylic acid and amide
Jun Liu, De-Xian Wang, Qi-Yu Zheng, Mei-Xiang Wang* Chin. J. Chem. 2006, 24, 1665-1668.
Nitrile biotransformations for highly enantioselective synthesis of oxiranecarboxamides with tertiary and quaternary stereocenters; efficient chemoenzymatic approaches to enantiopure α-methylated serine and isoserine derivatives
Mei-Xiang Wang,* Gang Deng, De-Xian Wang, Qi-Yu Zheng J. Org. Chem. 2005, 70, 2439-2444.
Synthesis of optically active α-methylamino acids and amides through biocatalytic kinetic resolution of amides
Mei-Xiang Wang,* Jun Liu, De-Xian Wang, Qi-Yu Zheng Tetrahedron: Asymmetry 2005, 16, 2409-2416.